 
The charge-to-mass ratio could then be calculated, and was found to be at least 1,000 times higher than that of the hydrogen atom.\): Millikan’s experiment measured the charge of individual oil drops. He is known for the Thomson atomic theory. tube Recommended PYQs (STRICTLY NCERT Based) Dual Nature of Radiation and Matter Physics Practice questions, MCQs, Past Year Questions (PYQs), NCERT. Thomson is credited with the discovery of the electron, the negatively charged particle in the atom. Thomson is buried in Westminster Abbey, near Sir Isaac Newton. Thomson, led to the discovery of the negatively charged part of the atom, the electron. An equal deflection was produced by applying a magnetic field, perpendicular to the electric field, using an electromagnet. He died August 30, 1940, Cambridge, Cambridgeshire, England. The cathode ray tube experiment, originally carried out by J.J. The beam was deflected by passing it through an electric field between the parallel plates.  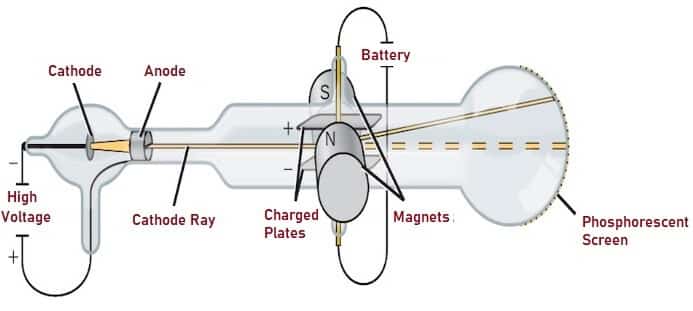
In the apparatus shown here, rays produced at the cathode were focused into a narrow beam by the slits in the cylinders in the narrow part of the tube, producing a fluorescent spot on the spherical end of the tube. The most diverse opinions are held as to these rays according to the almost unanimous opinion of German physicists they are due to. unobserved phenomena in the ther, of whose laws we are ignorant. By 1897, Thomson had measured the charge-to-mass ratio, but finding the charge or mass of the electron independently took a further two years. , 293-316 (1897) The experiments discussed in this paper were undertaken in the hope of gaining some information as to the nature of the Cathode Rays. Others, such as geophysicist Emil Wiechert, realized that the charged carriers of electricity were very small, but Thomson seems to have been the first to suggest that electrons - or 'corpuscles', as he called them - existed inside the atom he put forward a model for atomic structure that was based on the patterns of repulsion of floating magnets noted by Alfred Mayer in 1878. experiment to his assistants Hans Geiger and Ernest Marsden. First, in a variation of a pivotal 1895 experiment by Jean Perrin, he built a pair of cathode ray tubes ending in a pair of metal cylinders with a slit. JJ Thomson used his cathode ray experiment to disco. 
Three of his experiments proved especially conclusive. Thomson was an English physicist who worked with cathode ray tubes similar to those used by Crookes and others in the mid-19th century. This earlier generation had observed that these rays travelled in straight lines but could be deflected by a magnetic field, which became the basis of Thomson's work. Thomson refined previous experiments and designed new ones in his quest to uncover the true nature of these mysterious cathode rays. The discharge tubes and pumps invented by Heinrich Geissler in the 1850s, and the induction coil perfected by Heinrich Ruhmkorff at about the same time, had enabled many physicists, such as Julius Plücker and Johann Hittorf, to experiment with the new 'cathode rays'. Thomson found that the cathode rays can be deflected by an electric field, as shown below. The road leading to the discovery had been an arduous one, trodden by many scientists. A series of experiments with cathode rays he carried out near the end of the 19th century led to his discovery of the electron, a negatively charged atomic. Although it was certainly one of his instruments, he used a series of cathode-ray tubes in that year, so we cannot be certain.Īlthough the moment of discovery of the first subatomic particle has been controversial, Thomson's measurement of the charge-to-mass ratio for cathode rays is now considered to have defined the electron. British physicist (1856 1940) whose cathode ray tube experiment in 1897 showed that atoms were composed of smaller particles and led him to formulate the 'plum pudding' model of the atom. This has been claimed to be the apparatus with which Joseph John Thomson discovered the electron in 1897. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
